 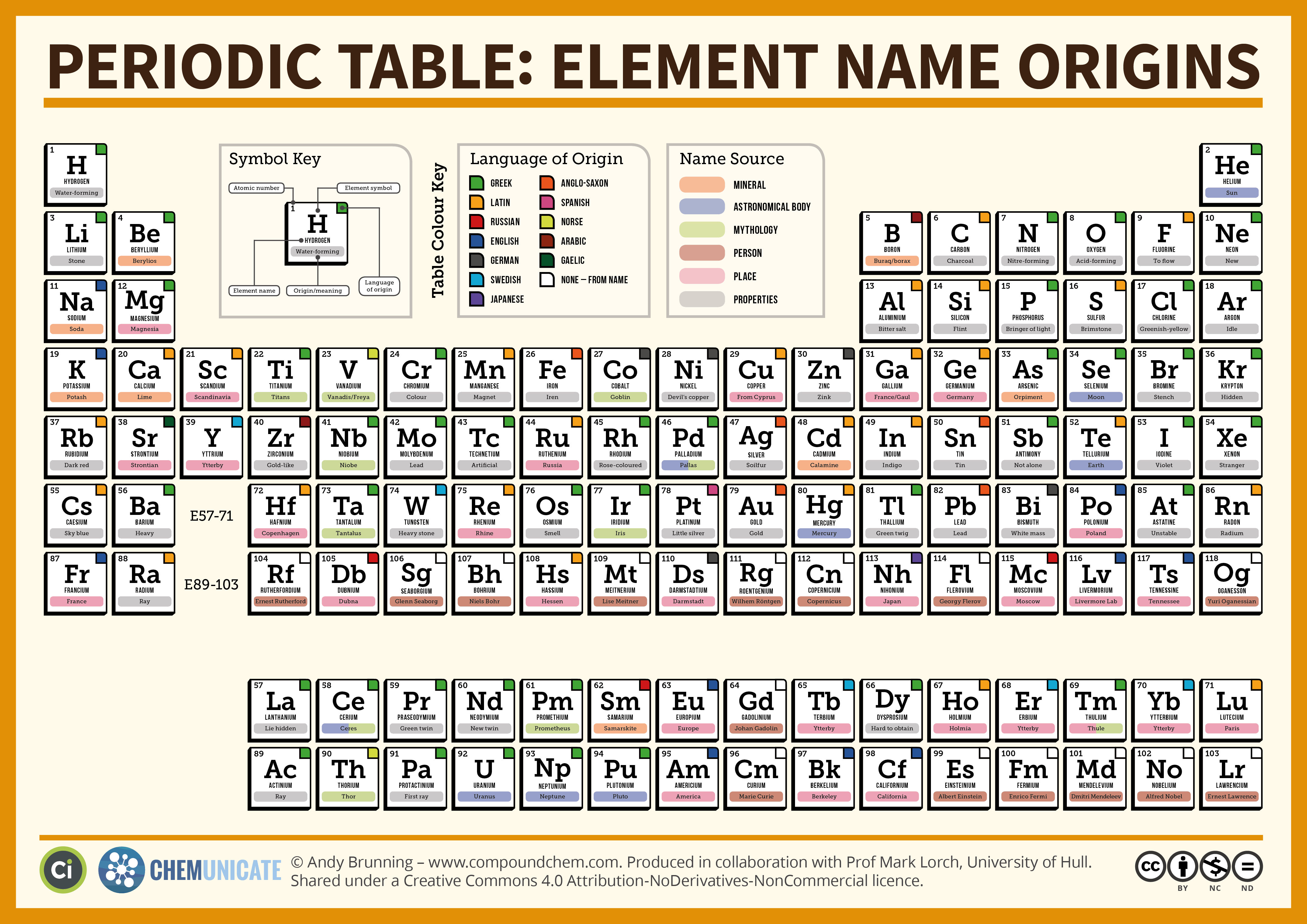
Nevertheless, it is possible for a vast majority of elements to form covalent molecules in which two like atoms are held together by a single covalent bond. Some are bound by covalent bonds in molecules, some are attracted to each other in ionic crystals, and others are held in metallic crystals. However, this idea is complicated by the fact that not all atoms are normally bound together in the same way. The atomic radius is one-half the distance between the nuclei of two atoms (just like a radius is half the diameter of a circle). This is caused by the increase in atomic radius. 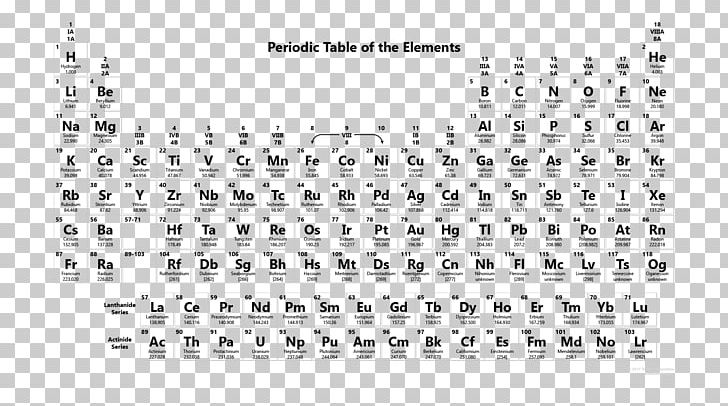
Electron affinity decreases from top to bottom within a group. 
This is caused by the decrease in atomic radius.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
